Lithium-chemistry batteries are replacing sealed lead-acid (SLA) and nickel metal-hydride (NiMH) types in many fixed and portable applications due to their higher energy storage density relative to both weight and volume. As larger lithium-chemistry batteries are designed, managing the waste heat generated by the ever-higher charge and discharge currents becomes an increasing challenge.
Excessive temperature rise in lithium-chemistry cell packs has always been a major design issue. Most lithium-ion (Li-ion) cells must not be charged above 45°C or discharged above 60°C. These limits can be pushed a bit higher, but at the expense of cycle life. In the worst case, if cell temperatures get too high, venting may occur, resulting in battery failure or even a cell fire. New lithium battery chemistries like lithium iron phosphate (LiFePO4) promise to increase both charge and discharge max temperatures, but there will always be a fairly low upper limit.
The waste heat energy that causes temperature rise in lithium-chemistry batteries comes from several sources. During both charge and discharge, electronic circuit elements located around the battery may conduct heat into the cells. This is especially true for chargers since they’re usually a switching power supply with a controller that implements the CC/CV algorithm required for the optimum charge of lithium-chemistry cells. At least 10% of the energy passed through such a charger is lost as waste heat, which can be conducted into the battery via terminals and other structures. Some charger architectures can have as low as 70% conversion efficiency.
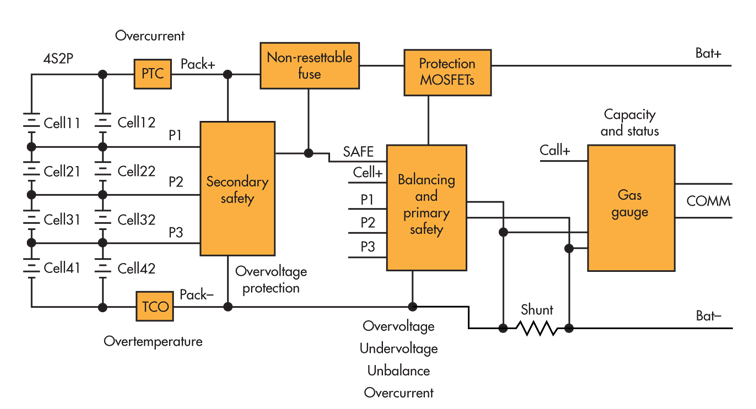
Other sources of waste heat include protection and gas-gauge circuits inside the battery itself. These heat sources include the positive temperature coefficient thermistor (PTC) and thermal cutoff fuse (TCO), electronically controlled fuse (usually a chemical self control protector), the primary protection MOSFETs, and the current measurement shunt for the gas gauge (Fig. 1). At high current, the resistance of the nickel strips used to interconnect the cells and circuit board traces also becomes significant.
 |
|
| Fig. 1. | Heat sources inside the battery include the PTC and TCO, the electronically controlled fuse, the primary protection MOSFETs, and the current measurement shunt for the gas gauge. |
Most electronic elements in the current path are resistive in nature. The heat produced is proportional to the square of the current through the element (P = R·I2). When charge and discharge currents are low (e.g., under 1 A), small resistive elements like the on resistance of protection MOSFETs and current shunt resistance produce little heat. However, these heat sources become critical design challenges as the current increases. It’s not unusual for large Li-ion batteries to deliver discharge current of 10 A or more and have charge current specs in the 5-A range. At these current levels, even the smallest resistive element can produce significant heat and contribute to battery temperature rise over a several-hour charge or discharge cycle.
A Look At The Cells
The electronics aside, an often neglected source of waste heat is the cells themselves. Li-ion cells feature overcurrent protection devices that are resistive and a source of heat when current is flowing. The anode and cathode of the cells conduct the current from the chemical reaction sites to the cell’s external contacts and have resistance that depends on their materials.
Most Li-ion cells have impedance specs in the 80- to 100-mΩ range and can be significant heat sources when the charge or discharge current is near the maximum for the cell. Cells are coming on the market with 10- to 20-C current ratings. (1 C is the capacity rating of the cell in A-hrs divided by 1 hour, so a 2400-mAh cell has a 1-C rating of 2.4 A.) Of course, the cell can’t sustain a 20-C current for very long, but it’s going to be fairly hot in the short time that it can unless it’s cooled in some way.
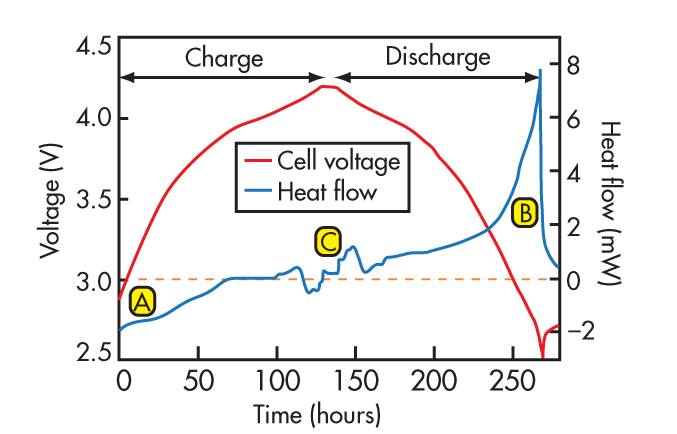
Almost completely ignored is the chemical reaction in the cell. The chemical reaction that takes place during the charging of a lithium-chemistry cell is endothermic, as it absorbs heat. Since there is no free lunch in thermodynamics, the discharge reaction is exothermic and produces heat. The Central Research Institute of Electric Power Industry (CRIEPI) conducted a classic study in 1995 that characterized these reactions in a calorimeter. Figure 2 was taken from a report on that research (see http://criepi.denken.or.jp/en/e_publication/a1996/96seika29.html).
 |
|
| Fig. 2. | In Li-ion cells, the charge chemical reaction is endothermic (A), and the discharge is exothermic. Note that near the end of the discharge, the heat produced increases rapidly, indicating a rapid increase in cell impedance near the end of cell capacity. |
This plot shows a charge cycle followed by a discharge cycle of a single Li-ion cell and details the heat flow into and out of the cell during this process. The initial section of the plot, labeled “A,” shows the endothermic nature of the charge chemical reaction. The discharge section, labeled “B,” is obviously exothermic. But the surprise is that near the end of discharge, the heat produced increases rapidly, indicating a rapid increase in cell impedance near the end of cell capacity. (Note that constant current charge and discharge was used.)
The endothermic nature of the charge chemical reaction is weak compared to other heat sources. In every case we’ve studied at Micro Power, battery temperature increased during charge because the other heat sources overwhelm the weak endothermic chemical reaction inside the cell.
The strong exothermic nature of the discharge chemical reaction may cause a large increase in temperature rise near the end of discharge. This is compounded by the fact that many times, the load on a battery is constant-power in nature. As the battery voltage decreases near the end of its capacity, the current must increase to maintain constant power. This causes all the resistive elements in the battery circuit to produce more heat and can result in a large battery temperature increase.
Elements To Include
A properly protected lithium-chemistry battery should have overtemperature protection built in. Most primary and secondary lithium-chemistry safety circuits include some provision to open the primary safety MOSFETs when the temperature is too high (and too low as well). As shown in Figure 1, some primary and secondary safety circuits can open an electronically controlled fuse as a last resort (because those fuses are usually not resettable and disable the pack when open).
There are many factors to consider when designing high-current lithium-chemistry batteries. One must design a method for removing waste heat from resistive circuit elements around the cells, and heat produced inside the cells themselves must be handled. At the high current levels found in transportation applications, active cooling with forced air or even circulating liquid must be used.
To avoid heating in the cells themselves, battery designers use several parallel columns of cells to reduce the current into and out of individual cells. However, this presents a challenge itself because a weak cell in one row can load the other cells in the same row, causing internal current flow in the cell array strapping. The proper placement of PTC devices can prevent this, but it adds cost and complexity.
As lithium-chemistry batteries get larger and are used in more high-current applications where SLA and nickel-chemistry batteries have dominated, battery designers must pay much more attention to heat sources and removal. The consequences of not designing for the heat that will be produced are at best a battery that doesn’t perform well and at worst a safety or reliability issue.
Related Articles